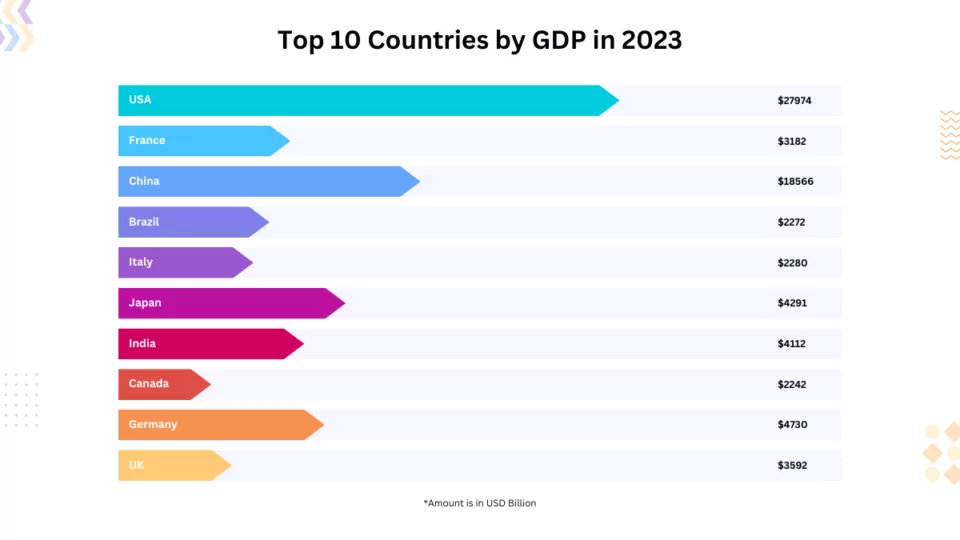
In today’s world, where fuel prices seem to rise with every passing day, owning a fuel-efficient car is more than just a luxury; it’s a necessity. Whether you’re commuting to work or planning a weekend getaway, having a car that goes the extra mile on a single tank of gas can save you a significant[……]