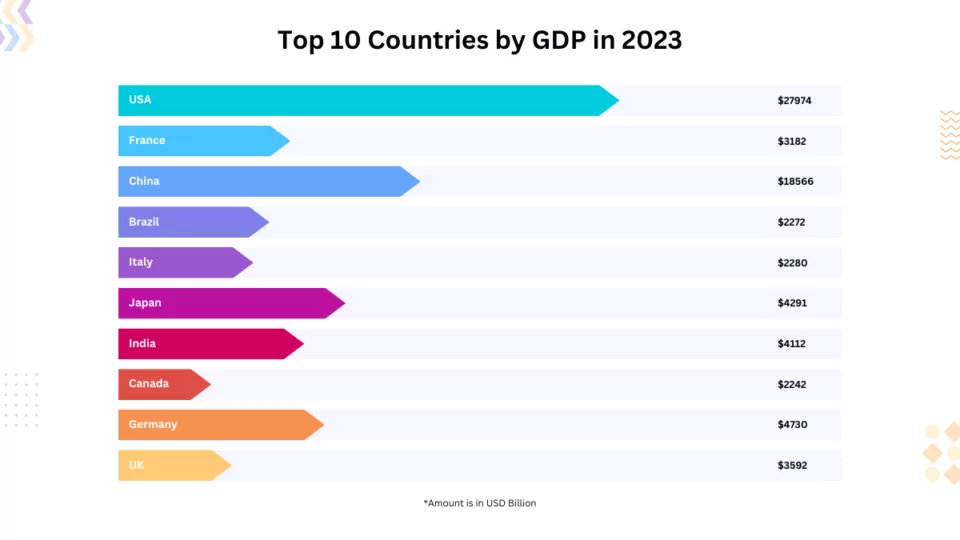
Introduction: Decoding the Top Model Kwid Car Price
Are you in the market for the latest and greatest deals on the top model Kwid car? Look no further! In this comprehensive guide, we’ll dive deep into the realm of the top model Kwid car price in 2024. From exploring the latest prices to uncovering[……]